 
Example: An example of dipole-dipole interaction is the interaction between two sulfur dioxide (SO2) molecules, where the sulfur atom of one molecule is attracted to the oxygen atoms of the other molecule. Since many molecules are polar, this is a common intermolecular force. If the molecule is symmetrical, then there is no dipole because the dipole moments won’t cancel out. Symmetry of the compound indicates whether there is a dipole moment for the compound. Add each dipole moment of each bond to form an overall dipole moment for the molecule. How do you determine the dipole moment?ĭetermine the overall dipole of the compound. (Permanent dipoles are found in the dipole-dipole forces.) an induced dipole in a non polar molecule : The London dispersion force is a temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles. What is the difference between a permanent molecular dipole in a polar molecule and an induced dipole in a non polar molecule? This uneven distribution of electrons can make one side of the atom more negatively charged than the other, thus creating a temporary dipole, even on a non-polar molecule. Temporary dipoles are created when electrons, which are in constant movement around the nucleus, spontaneously come into close proximity. The only intermolecular forces in methane are London dispersion forces. These are called London dispersion forces and they are the weakest intermolecular force as the dipoles are only temporary. In contrast, permanent dipole refers to the dipole moment that originally occurs in a compound due to uneven electron distribution. Summary – Induced Dipole vs Permanent Dipole Induced dipole refers to the dipole moment that creates in a nonpolar compound due to the effect of an ion nearby. What is the difference between dipole moment and permanent dipole moment? Intramolecular forces are stronger than intermolecular forces. Hydrogen Bonding are a special type of permanent dipole – permanent dipole forces. Permanent dipole – dipole forces are the attractive forces between two neighbouring molecules with a permanent dipole. What is a permanent dipole a level chemistry? PH3 has a lone pair and does not have a trigonal planar geometry–for this reason it is not symmetrical. PH3 must be polar since it is not symmetrical. It forms dipole-dipole because it is a polar molecule. Induced (temporary) dipole moments are created when an external electric field distorts the electron cloud of a neutral molecule. There are two kinds of dipole moments: Permanent electric dipole moments can arise when bonding occurs between elements of differing electronegativities. What is the difference between temporary dipole and permanent dipole? This force is sometimes called an induced dipole-induced dipole attraction. The London dispersion force is a temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles. As a result, oxygen has a slight negative charge (δ-). The 10 electrons of a water molecule are found more regularly near the oxygen atom’s nucleus, which contains 8 protons. oxygen is more electronegative than hydrogen). The permanent dipole in water is caused by oxygen’s tendency to draw electrons to itself (i.e. A molecule with a permanent dipole moment is called a polar molecule. These occur when two atoms in a molecule have substantially different electronegativity: One atom attracts electrons more than another, becoming more negative, while the other atom becomes more positive. This type of intermolecular bond is stronger than London dispersion forces with the same number of electrons. Polar molecules display attractions between the oppositely charged ends of the molecules. Permanent dipole-permanent dipole interactions Molecules with a permanent dipole are polar. 7 How do you determine the dipole moment?.3 What is the difference between temporary dipole and permanent dipole?. 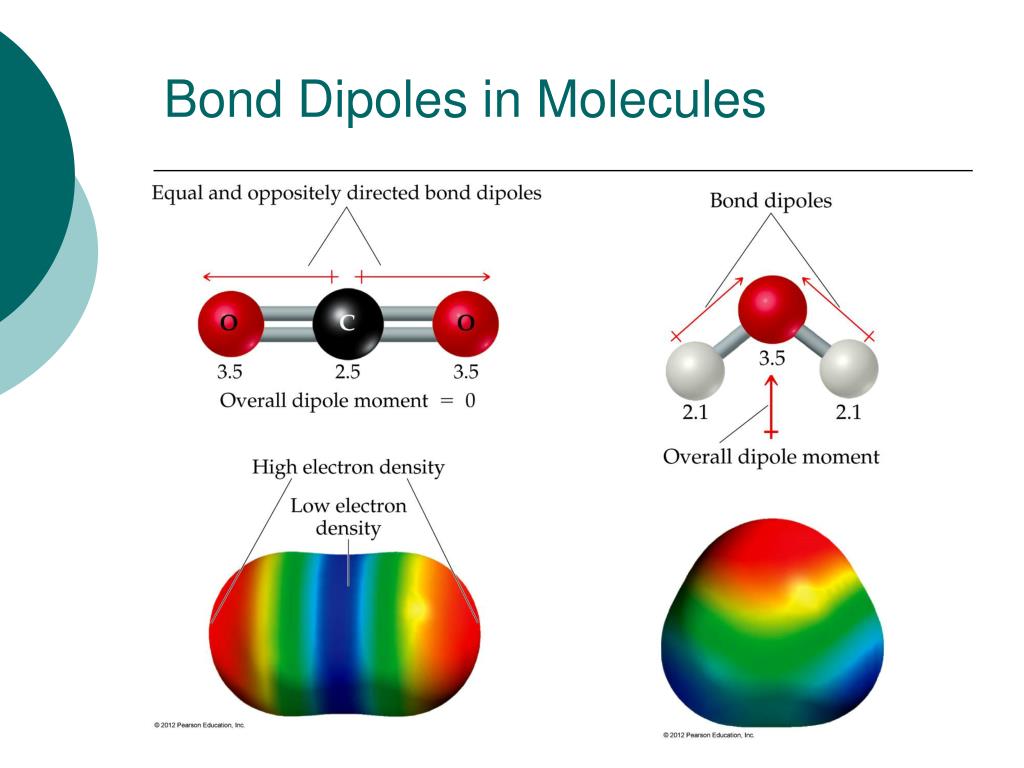
1 What is permanent dipole dipole forces?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
